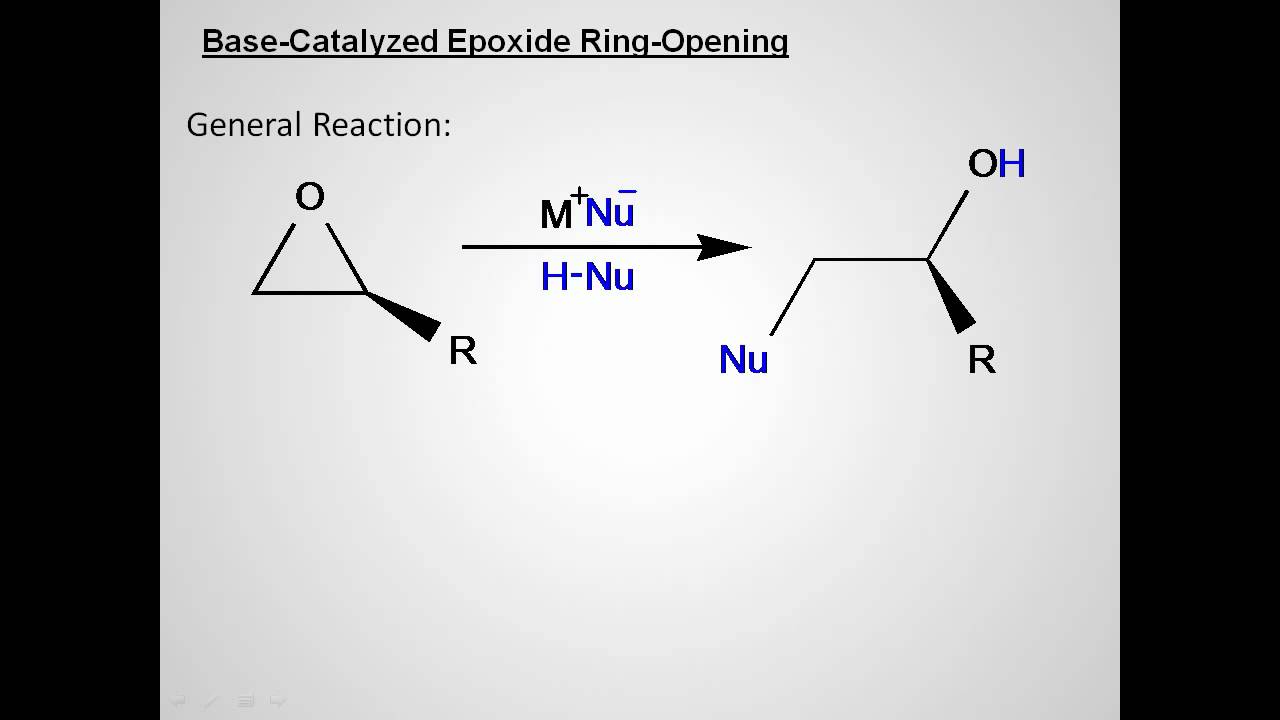
Then the carbon-oxygen bond begins to break (step 2) and positive charge begins to build up on the more substituted carbon (recall the discussion from section 8.4B about carbocation stability). First, the oxygen is protonated, creating a good leaving group (step 1 below). Lab 4 - Ring Opening of an Epoxide Lab Report for the ring-opening of 1,2-epoxyhexane under both acidic and basic conditions. Probably the best way to depict the acid-catalyzed epoxide ring-opening reaction is as a hybrid, or cross, between an S N2 and S N1 mechanism. Like in other S N2 reactions, nucleophilic attack takes place from the backside, resulting in inversion at the electrophilic carbon. Immobilization of functional molecules in ordered microarrays has vast importance for many applications in sensing and diagnostics. So in this video, well look at the ring opening reactions of epoxides using strong nucleophiles. This accounts for the observed regiochemical outcome. If oxides have a lot of ring or angle strain, this makes them very reactive towards ring opening. What about the electrophile? There are two electrophilic carbons in the epoxide, but the best target for the nucleophile in an S N2 reaction is the carbon that is least hindered. Ring-opening reactions can proceed by either S N 2 or S N 1 mechanisms, depending on the nature of the epoxide and on the reaction conditions. When a nucleophilic substitution reaction involves a poor leaving group and a powerful nucleophile, it is very likely to proceed by an S N2 mechanism. The nonenzymatic ring-opening reactions of epoxides provide a nice overview of many of the concepts we have seen already in this chapter. 
Although significant progress has been made, the established methods were limited to titanocene-catalyzed reactions. The use of 1,2-epoxyhexane and 1,2-epoxydodecane as substrates gave both regioisomers in. The reductive ring opening of epoxides is a powerful transformation to convert readily accessible epoxides into a diverse array of valuable alcohols, including pharmaceuticals, agrochemicals, and functional polymers. Generally, the nucleophilicity is enhanced with the increase of cation radius and anion branches.The nucleophile itself is potent: a deprotonated, negatively charged methoxide ion. Like glycidol, however, the product epoxides are susceptible to ring opening via nucleophilic attack at the C-3 position. Control of regio- and stereoselectivity of chemical reactions is the central theme in synthetic chemistry. The ring opening of cyclic epoxides gave 77-97 yields of trans-beta-methoxy alcohols, in 0.5-6 h. It was found that the alkalinity of the carboxylate ions and the electrostatic interaction are the key factors to affect the nucleophilicity of catalysts. in this paper, we report a metal- and solvent-free ring-opening procedure of symmetrical and unsymmetrical epoxides with various aromatic and aliphatic amines. The influences of solvent environment, as well as the structure and properties of catalysts on their activity were further studied. And the nucleophile is going to end up opening the ring and adding, in an anti fashion, to the OH that is created here. And in the second step, we add a proton source. The epoxy ring opening of 20 with 2-lithio-2-methylpropionitrile, derived from isobutyronitrile using LDA as a base, afforded an approximately 1:1 mixture of heptanenitrile 31 and a five-membered cyclic imidate 32. So in the first step, we add a strong nucleophile to our epoxide. The reaction orders of reactants and catalyst were obtained by kinetic experiments, and based on that, the kinetic equation and mechanism were put forward. Oxirane ring opening is a synthetically significant reaction for the creation of new pharmaceutically important scaffolds. So in this video, we'll look at the ring opening reactions of epoxides using strong nucleophiles. In this work, the carboxylate catalysts were directly prepared rather than using the traditional catalysts like amines or quaternary ammonium salts, thus removing the induction period and making the kinetic results more accurate and rigorous. However, the systematical study on the structure-activity relation of catalysts is still scarce. The ring-opening reactions of epoxides with carboxylic acids have great applications in the coating and polymer industry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
